Italy seeks volunteers for Covid-19 vaccine trial
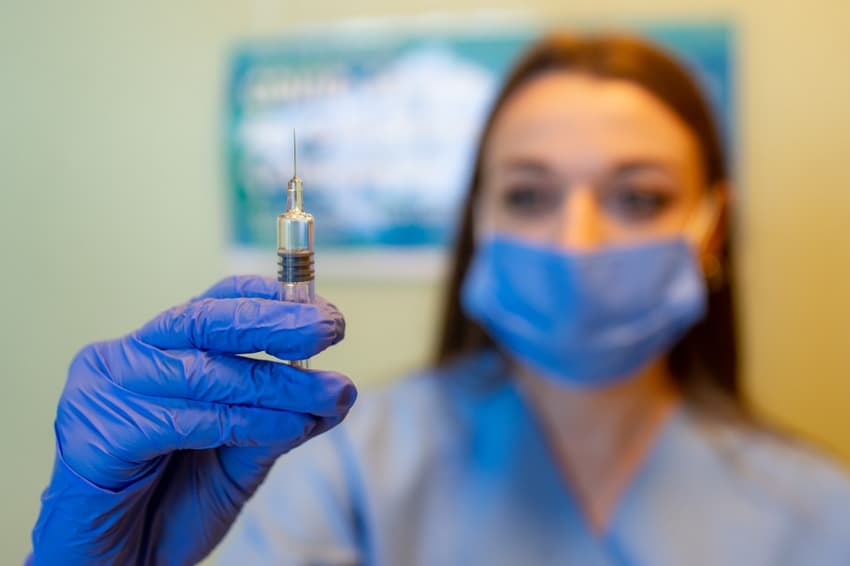
Italian researchers hope to test a potential vaccine against the new coronavirus on 300 volunteers, starting as soon as next month.
Volunteers in Italy could receive the first doses in December, as scientists begin a Phase 3 clinical trial of the vaccine developed by Oxford University and AstraZeneca pharmaceutical group and partly manufactured and bottled by two Italian companies near Rome.
The vaccine, one of several in development around the world, is among the most advanced, with a large-scale trial already underway on as many as 10,000 people in the UK.
READ ALSO: What you need to know about getting a flu vaccination in Italy this year
Researchers are seeking 300 volunteers for the two-year Italian trial, which will be carried out by seven institutions starting with the University Hospital of Modena.
Though one recent study suggested that nearly one in two people in Italy would be 'hesitant' about getting a new Covid-19 vaccine, the hospital has already been inundated with inquiries from people interested in taking part, head of the infectious diseases department Cristina Mussini told reporters this week.
The hospital plans to set up a dedicated phone hotline to collect applications throughout November, with spots going to the first people who volunteer and meet the selection criteria.

Doses of the Oxford vaccine at a facility near Rome, where Italian company Catalent Biologics is responsible for packaging it. Photo: Vincenzo Pinto/AFP
Participants should be over 18, not pregnant or immunosuppressed, and should not have already contracted a severe case of Covid-19.
Only 200 of them will receive the experimental vaccine, with the remaining 100 getting a placebo. Neither subjects nor researchers will be informed which one each volunteer gets until the study is complete.
Participants will receive two doses and be subjected to periodic blood and health checks over a total of two years, though the first results of the study are expected to be announced after six months.
Phase 3 trials are the final tests before regulators decide whether to approve a drug. The European Medicines Agency, which reviews drugs for use within the European Union, hopes to fast-track approval for Covid-19 vaccines, and the head of Italy's Higher Health Council, Franco Locatelli, has said the first doses could be available in spring 2021.
The new trial is separate from an early-stage trial underway in Rome, where researchers at the Spallanzani National Institute for Infectious Diseases are testing a different vaccine developed by Italian biotech company ReiThera on a much smaller sample of volunteers.
How does the vaccine work?
Officially known as ChAdOx1 nCoV-19 or AZD1222, the Oxford vaccine works by targeting a spiky structure on the surface of the coronavirus called the S protein, which it uses to attach to human cells and cause an infection.

A model of the SARS-CoV-2 virus. Photo: Jens Schlueter/AFP
The genetic material of the new virus's S protein is placed in a weakened version of a common cold virus from chimpanzees that has been modified to prevent it being able to replicate in humans, so that after injection copies of only the S protein (not the virus) are produced.
The idea is that the body will detect the S protein and develop an immune response, teaching the immune system to attack S proteins in future. If the new virus enters a vaccinated person's body, scientists hope the immune system would target its surface spikes, thereby helping to prevent it binding to cells and reproducing.
"A significant proportion of vaccines that are tested in clinical trials don't work," warns the Oxford Vaccine Centre, which developed the vaccine with Oxford University’s Jenner Institute.
"If we are unable to show that the vaccine is protective against the virus, we would review progress, examine alternative approaches, such as using different numbers of doses, and would potentially stop the programme."
Comments
See Also
Volunteers in Italy could receive the first doses in December, as scientists begin a Phase 3 clinical trial of the vaccine developed by Oxford University and AstraZeneca pharmaceutical group and partly manufactured and bottled by two Italian companies near Rome.
The vaccine, one of several in development around the world, is among the most advanced, with a large-scale trial already underway on as many as 10,000 people in the UK.
READ ALSO: What you need to know about getting a flu vaccination in Italy this year
Researchers are seeking 300 volunteers for the two-year Italian trial, which will be carried out by seven institutions starting with the University Hospital of Modena.
Though one recent study suggested that nearly one in two people in Italy would be 'hesitant' about getting a new Covid-19 vaccine, the hospital has already been inundated with inquiries from people interested in taking part, head of the infectious diseases department Cristina Mussini told reporters this week.
The hospital plans to set up a dedicated phone hotline to collect applications throughout November, with spots going to the first people who volunteer and meet the selection criteria.

Doses of the Oxford vaccine at a facility near Rome, where Italian company Catalent Biologics is responsible for packaging it. Photo: Vincenzo Pinto/AFP
Participants should be over 18, not pregnant or immunosuppressed, and should not have already contracted a severe case of Covid-19.
Only 200 of them will receive the experimental vaccine, with the remaining 100 getting a placebo. Neither subjects nor researchers will be informed which one each volunteer gets until the study is complete.
Participants will receive two doses and be subjected to periodic blood and health checks over a total of two years, though the first results of the study are expected to be announced after six months.
Phase 3 trials are the final tests before regulators decide whether to approve a drug. The European Medicines Agency, which reviews drugs for use within the European Union, hopes to fast-track approval for Covid-19 vaccines, and the head of Italy's Higher Health Council, Franco Locatelli, has said the first doses could be available in spring 2021.
The new trial is separate from an early-stage trial underway in Rome, where researchers at the Spallanzani National Institute for Infectious Diseases are testing a different vaccine developed by Italian biotech company ReiThera on a much smaller sample of volunteers.
How does the vaccine work?
Officially known as ChAdOx1 nCoV-19 or AZD1222, the Oxford vaccine works by targeting a spiky structure on the surface of the coronavirus called the S protein, which it uses to attach to human cells and cause an infection.

A model of the SARS-CoV-2 virus. Photo: Jens Schlueter/AFP
The genetic material of the new virus's S protein is placed in a weakened version of a common cold virus from chimpanzees that has been modified to prevent it being able to replicate in humans, so that after injection copies of only the S protein (not the virus) are produced.
The idea is that the body will detect the S protein and develop an immune response, teaching the immune system to attack S proteins in future. If the new virus enters a vaccinated person's body, scientists hope the immune system would target its surface spikes, thereby helping to prevent it binding to cells and reproducing.
"A significant proportion of vaccines that are tested in clinical trials don't work," warns the Oxford Vaccine Centre, which developed the vaccine with Oxford University’s Jenner Institute.
"If we are unable to show that the vaccine is protective against the virus, we would review progress, examine alternative approaches, such as using different numbers of doses, and would potentially stop the programme."
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.