For more information about the current coronavirus-related restrictions on travel to Italy please see the Foreign Ministry’s website (in English).
Reader questions
For Members
Reader question: When will Italy recognise the Indian-produced AstraZeneca vaccine?
Various countries across the EU have announced approval of the Indian-produced batches of AstraZeneca, known as Covishield, allowing quarantine-free travel. With Italy yet to follow, many readers immunised with this jab have contacted The Local to ask how they can travel to Italy. Here's what you need to know.
Question: We were vaccinated with the AstraZeneca jabs that were produced in India, called Covishield. We would like to travel to Italy but we heard these particular batches won't be accepted there. What can we do?
Italy opened up its borders to vaccinated travellers for the summer back in May in accordance with the European Medicines Agency (EMA) approved vaccines list.
Travel was re-introduced under the EU's 'health pass' scheme, allowing people to travel within the bloc quarantine-free based on proof that you have either been fully vaccinated, have recently tested negative or have antibodies after recovering from Covid-19.
Proof of being fully immunised is currently recognised if you've had one of the following EMA-authorised vaccines:
- Spikevax (previously Moderna)
- Pfizer-BioNTech (also called Comirnaty)
- Vaxzevria (also called AstraZeneca)
- Johnson & Johnson (also called Janssen)
Showing evidence of the Pfizer, UK-made AstraZeneca, Johnson & Johnson and Moderna vaccines allows travellers to come to Italy without quarantining on arrival, although there are exceptions to this, such as for those arriving from the UK who still have to complete a five-day quarantine.
However, many travellers have found that even though they were vaccinated with the AstraZeneca vaccine, they are blocked from entering Italy via proof of vaccination - because the country still doesn't recognise three batches that were produced by the Serum Institute of India (SII), known as Covishield.
Italy still has made no announcement of when - or if - it will begin to recognise the Indian-made AstraZeneca jab.
EU member states can allow entry for people vaccinated with jabs enrolled on the World Health Organisation’s Emergency Use Listing process, which Covishield is, but they can decide not to recognise the vaccine until it is approved by the EMA - which it never did.
And so Italy never did either.
The Covishield vaccine has "not yet been approved by the EMA and are therefore not recognised by the Italian authorities," stated the Italian consulate in Mumbai.
Would-be holidaymakers took to Twitter last month to express their outrage over upended travel plans. Although the UK's Prime Minister Boris Johnson told reporters there will "not prove to be a problem" for travellers who have received one of these jabs.
https://twitter.com/queenchristina_/status/1410996822718431238
Not only that - since Italy expanded its green pass as a mandatory requirement to access most public sites and venues, people are required to show proof of vaccination, testing or recovery across most of the country after arrival too.
What's the difference between Vaxzevria and Covishield?
The AstraZeneca Covid-19 vaccine was first created in the UK by Oxford University and British-Swedish pharmaceutical company, AstraZeneca, and the UK-made version has been identified and sold as Vaxzevria.
Some batches were manufactured in India and identified as Covishield.
The UK received some five million doses of the AstraZeneca vaccine from the Serum Institute back in March.
There is no difference between the two AstraZeneca vaccines - only that the EMA is yet to approve the one made by the Serum Institute of India.
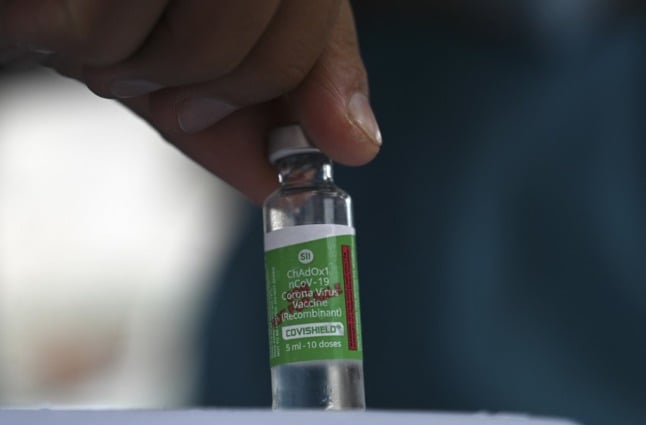
Photo by TAUSEEF MUSTAFA / AFP
How do I know if my AstraZeneca jab was one of the Covishield batches?
If you received an AstraZeneca vaccine dose, you can find out which batch your jab came from by looking on your vaccination card – the batch number is listed alongside the type of vaccine you received and the date of your vaccination.
The numbers of the Indian-produced AstraZeneca batches still not currently approved by the EMA are 4120Z001, 4120Z002 and 4120Z003.
NHS app users in England and Wales can check their batch number on the app.
Canadian travellers can check their personal immunisation records.
American travellers would need to look on their CDC vaccination cards.
Despite Italy not recognising Covishield as proof of immunisation, one reader who received one shot of an Indian-made batch of AstraZeneca told The Local her proof of vaccination was accepted when she travelled to Italy in July.

She was vaccinated in the UK with one Covishield jab and the second dose was the British-manufactured AstraZeneca.
However, both vaccines are labelled as 'Vaxzevria' as they were both administered in the UK, in accordance with guidelines set out by the UK's Department of Health and Social Care (DHSC).
So only by reading the batch numbers can you tell the origin.
Whether not having Covishield specifically written on your document would be accepted for travellers from other destinations such as the US, Canada and Australia is unclear, as UK arrivals must quarantine anyway.
READ ALSO:
- Q&A: Your questions about Italy’s quarantine for UK arrivals answered
- What you need to know about travel between Italy and Canada
- UK changes travel rules again to impose quarantine on European arrivals who had mixed vaccine doses
What can I do if Italy doesn't approve Covishield?
Italy confirmed it would recognise Covid health certificates from five non-EU countries last month, including Canada, Israel, Japan, the UK and the US.
While visitors from Canada, Israel, Japan and the US can already use health certificates issued in their own countries to avoid quarantine in Italy, the Italian government continues to impose separate restrictions on the UK that oblige travellers to self-isolate for five days on arrival until August 30th.
For details on which countries need to quarantine and those who can avoid it, check here.
If you've been vaccinated with Covishield, your other two options are testing or showing proof of recovery.
And once you're in Italy, this still holds true. If you can't show proof of having recovered from the virus within the previous six months, you'll need to keep getting tested in order to access most of Italy's cultural sites and leisure venues including indoor restaurants, museums, concert venues, gyms, spas, theme parks and much more.
EXPLAINED:
- How can you get Italy’s ‘green pass’ if you’re not vaccinated?
- What are the Covid-19 testing rules for travelling between the US and Italy?
Do other EU countries recognise the Covishield batches?
Other countries in the EU and Schengen area have approved the Indian-produced vaccine, including France, which announced last month that travellers immunised with Covishield could enter the country without quarantining.
The EU countries that currently accept the vaccine produced in India are: Austria, Belgium, Bulgaria, Finland, Germany, Greece, Hungary, Iceland, Ireland, Slovenia, the Netherlands, France, Romania, Spain and Sweden, with the addition of Switzerland outside the EU.
The Local will continue to follow any updates to the travel restrictions closely. Please check our homepage or travel news section for the most recent reports on any changes
Comments (3)
See Also
Question: We were vaccinated with the AstraZeneca jabs that were produced in India, called Covishield. We would like to travel to Italy but we heard these particular batches won't be accepted there. What can we do?
Italy opened up its borders to vaccinated travellers for the summer back in May in accordance with the European Medicines Agency (EMA) approved vaccines list.
Travel was re-introduced under the EU's 'health pass' scheme, allowing people to travel within the bloc quarantine-free based on proof that you have either been fully vaccinated, have recently tested negative or have antibodies after recovering from Covid-19.
Proof of being fully immunised is currently recognised if you've had one of the following EMA-authorised vaccines:
- Spikevax (previously Moderna)
- Pfizer-BioNTech (also called Comirnaty)
- Vaxzevria (also called AstraZeneca)
- Johnson & Johnson (also called Janssen)
Showing evidence of the Pfizer, UK-made AstraZeneca, Johnson & Johnson and Moderna vaccines allows travellers to come to Italy without quarantining on arrival, although there are exceptions to this, such as for those arriving from the UK who still have to complete a five-day quarantine.
However, many travellers have found that even though they were vaccinated with the AstraZeneca vaccine, they are blocked from entering Italy via proof of vaccination - because the country still doesn't recognise three batches that were produced by the Serum Institute of India (SII), known as Covishield.
Italy still has made no announcement of when - or if - it will begin to recognise the Indian-made AstraZeneca jab.
EU member states can allow entry for people vaccinated with jabs enrolled on the World Health Organisation’s Emergency Use Listing process, which Covishield is, but they can decide not to recognise the vaccine until it is approved by the EMA - which it never did.
And so Italy never did either.
The Covishield vaccine has "not yet been approved by the EMA and are therefore not recognised by the Italian authorities," stated the Italian consulate in Mumbai.
Would-be holidaymakers took to Twitter last month to express their outrage over upended travel plans. Although the UK's Prime Minister Boris Johnson told reporters there will "not prove to be a problem" for travellers who have received one of these jabs.
https://twitter.com/queenchristina_/status/1410996822718431238
Not only that - since Italy expanded its green pass as a mandatory requirement to access most public sites and venues, people are required to show proof of vaccination, testing or recovery across most of the country after arrival too.
What's the difference between Vaxzevria and Covishield?
The AstraZeneca Covid-19 vaccine was first created in the UK by Oxford University and British-Swedish pharmaceutical company, AstraZeneca, and the UK-made version has been identified and sold as Vaxzevria.
Some batches were manufactured in India and identified as Covishield.
The UK received some five million doses of the AstraZeneca vaccine from the Serum Institute back in March.
There is no difference between the two AstraZeneca vaccines - only that the EMA is yet to approve the one made by the Serum Institute of India.

Photo by TAUSEEF MUSTAFA / AFP
How do I know if my AstraZeneca jab was one of the Covishield batches?
If you received an AstraZeneca vaccine dose, you can find out which batch your jab came from by looking on your vaccination card – the batch number is listed alongside the type of vaccine you received and the date of your vaccination.
The numbers of the Indian-produced AstraZeneca batches still not currently approved by the EMA are 4120Z001, 4120Z002 and 4120Z003.
NHS app users in England and Wales can check their batch number on the app.
Canadian travellers can check their personal immunisation records.
American travellers would need to look on their CDC vaccination cards.
Despite Italy not recognising Covishield as proof of immunisation, one reader who received one shot of an Indian-made batch of AstraZeneca told The Local her proof of vaccination was accepted when she travelled to Italy in July.

She was vaccinated in the UK with one Covishield jab and the second dose was the British-manufactured AstraZeneca.
However, both vaccines are labelled as 'Vaxzevria' as they were both administered in the UK, in accordance with guidelines set out by the UK's Department of Health and Social Care (DHSC).
So only by reading the batch numbers can you tell the origin.
Whether not having Covishield specifically written on your document would be accepted for travellers from other destinations such as the US, Canada and Australia is unclear, as UK arrivals must quarantine anyway.
READ ALSO:
- Q&A: Your questions about Italy’s quarantine for UK arrivals answered
- What you need to know about travel between Italy and Canada
- UK changes travel rules again to impose quarantine on European arrivals who had mixed vaccine doses
What can I do if Italy doesn't approve Covishield?
Italy confirmed it would recognise Covid health certificates from five non-EU countries last month, including Canada, Israel, Japan, the UK and the US.
While visitors from Canada, Israel, Japan and the US can already use health certificates issued in their own countries to avoid quarantine in Italy, the Italian government continues to impose separate restrictions on the UK that oblige travellers to self-isolate for five days on arrival until August 30th.
For details on which countries need to quarantine and those who can avoid it, check here.
If you've been vaccinated with Covishield, your other two options are testing or showing proof of recovery.
And once you're in Italy, this still holds true. If you can't show proof of having recovered from the virus within the previous six months, you'll need to keep getting tested in order to access most of Italy's cultural sites and leisure venues including indoor restaurants, museums, concert venues, gyms, spas, theme parks and much more.
EXPLAINED:
- How can you get Italy’s ‘green pass’ if you’re not vaccinated?
- What are the Covid-19 testing rules for travelling between the US and Italy?
Do other EU countries recognise the Covishield batches?
Other countries in the EU and Schengen area have approved the Indian-produced vaccine, including France, which announced last month that travellers immunised with Covishield could enter the country without quarantining.
The EU countries that currently accept the vaccine produced in India are: Austria, Belgium, Bulgaria, Finland, Germany, Greece, Hungary, Iceland, Ireland, Slovenia, the Netherlands, France, Romania, Spain and Sweden, with the addition of Switzerland outside the EU.
The Local will continue to follow any updates to the travel restrictions closely. Please check our homepage or travel news section for the most recent reports on any changes
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.