Italy approves 'mix and match' vaccinations for under-60s as regions issue varying rules on AstraZeneca
The Italian Medicines Agency (Aifa) on Tuesday approved giving people under 60 a combination of Covid-19 vaccines after the government limited use of the AstraZeneca jab.
The health ministry said anyone under 60 who had already received a first dose of AstraZeneca should now have Pfizer or Moderna - so-called mRNA vaccines - for the second dose.
READ ALSO: Italy limits use of AstraZeneca vaccines to over-60s
Aifa on Monday published its approval for mixed vaccines. The regulator had previously recommended that people get both doses of the same vaccine.
The health ministry stated on Saturday that regional health authorities should stop giving the AstraZeneca jab to under-60s, amid concerns about heightened health risks for younger people.
Italian authorities had previously recommended AstraZeneca be only given to over-60s, but this was not an outright ban and many regions had been giving it to younger people on a voluntary basis. This advice was strengthened after the latest review last week.
However, despite the health ministry's new instructions, Italy’s regions are continuing to implement their own vaccine programmes with varying rules.
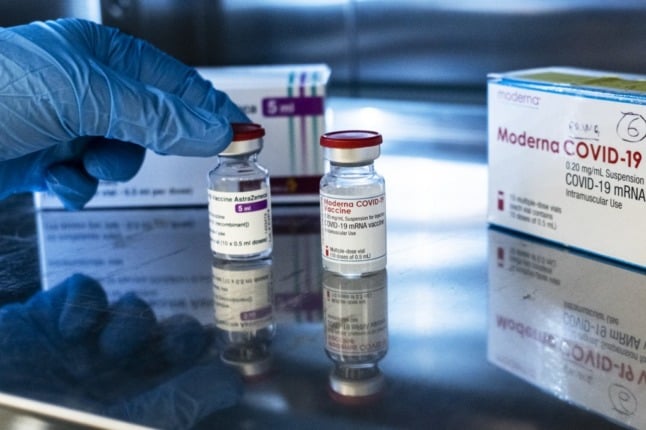
Vials of the AstraZeneca (L) and Moderna Covid-19 vaccines at the pharmacy of the Sant'Andrea hospital in Vercelli, Piedmont. Photo: MARCO BERTORELLO/AFP
Italian news agency Ansa reported “chaos in the regions” on Tuesday, saying the latest rule-change on AstraZeneca had "caused confusion”.
Some Italian regional authorities said they will still continue to allow people to take AstraZeneca if they want to amid concerns about the supply of other vaccines, while Campania said it was banning the use of the Astrazeneca and Johnson and Johnson vaccines altogether.
EXPLAINED: Why has Italy recommended the AstraZeneca vaccine for over-60s only?
Vincenzo De Luca, regional president of Campania, announced that he had written to the health minister saying his region was against giving mixed vaccinations and claiming that the practice "has not been trialled broadly on an international level".
NO AL MIX DI VACCINI
NO ALLA PRIMA DOSE DI VACCINI A VETTORE VIRALE
Ecco la mia dichiarazione 👇https://t.co/2yih6a3A1g
— Vincenzo De Luca (@VincenzoDeLuca) June 13, 2021
In an interview with La Stampa on Monday, Health Minister Roberto Speranza stressed that mixing vaccinations was known to be safe and effective, saying: "Our indications are peremptory and must be followed. It is not a political debate. It’s not the prime minister, ministers or a regional president who decides:”
"So-called 'vaccination crossing' is something Germany has been doing for two months, which France and Spain have also been doing for some time,” he said. “It's not made up, these are scientific studies. This is a procedure that has given good results.”
Several other EU countries including Sweden, Norway and Austria are also allowing people to have an mRNA shot for their second dose if they were first vaccinated with AstraZeneca, after those countries also changed their advice on the jab.
READ ALSO: Is Italy really going to offer vaccines to tourists this summer?
The European Medicines Agency (EMA) has stressed that the AstraZeneca jab remains approved for all adults, saying the benefits continue to outweigh the risks.
Misinformation is making the rounds today. This is the situation: Benefit/risk balance of AstraZeneca #COVID19 vaccine is positive and it remains authorised for all populations.
— EU Medicines Agency (@EMA_News) June 13, 2021
Meanwhile the Italian region of Puglia said it would allow people to take AstraZeneca if approved by their doctor, and Lombardy’s regional government expressed concerns about the supplies of Pfizer and Moderna vaccines.
Several regions have found themselves with unused doses of the Oxford University vaccine, as people opt to get vaccinated with alternatives that they perceive as safer or that have a shorter interval between doses (or in the case of Johnson & Johnson, require just one shot).
As a result, some regions had been offering the Covid-19 vaccine to younger adults as part of vaccination “open days”: special vaccination drives, often at evenings or weekends, that allow the youngest age groups to get a shot earlier than they otherwise might.
The latest review of the use of AstraZeneca in Italy came following the news that an 18-year-old in north-west Italy who was vaccinated with AstraZeneca two weeks previously had died after developing a blood clot.
Most of the AstraZeneca side effects observed so far occurred within two weeks of the first dose. The second dose is thought to carry an even lower risk, though more data is needed to know for sure.
Data from millions of people vaccinated with AstraZeneca across Europe and the UK indicates that serious side effects after either dose remain extremely rare.
In a monitoring report released this week, AIFA said that in Italy the incidence of blood clots after vaccination was around one per 100,000 injections of AstraZeneca, mainly in people under 60.
It has not received reports of any clots developing after the second dose, the agency said.
What are the different types of Covid-19 vaccine available?
AstraZeneca Johnson & Johnson, both so-called 'viral vector' vaccines, are authorised for everyone over 18 in Italy and by Europe’s medicines agency, but both have been dogged by reports of rare blood clots.
The EU and Italy have also authorised two mRNA vaccines: Pfizer/BioNTech and Moderna.
Viral vector vaccines like AstraZeneca and Johnson & Johnson use genetically-engineered version of a common-cold causing adenovirus as a "vector" to shuttle genetic instructions into human cells.
The mRNA or Messenger RNA genetic technology instead trains the body to reproduce spike proteins, similar to that found on the coronavirus.
When exposed to the real virus later, the body recognises the spike proteins and is able to fight them off.
Comments (1)
See Also
The health ministry said anyone under 60 who had already received a first dose of AstraZeneca should now have Pfizer or Moderna - so-called mRNA vaccines - for the second dose.
READ ALSO: Italy limits use of AstraZeneca vaccines to over-60s
Aifa on Monday published its approval for mixed vaccines. The regulator had previously recommended that people get both doses of the same vaccine.
The health ministry stated on Saturday that regional health authorities should stop giving the AstraZeneca jab to under-60s, amid concerns about heightened health risks for younger people.
Italian authorities had previously recommended AstraZeneca be only given to over-60s, but this was not an outright ban and many regions had been giving it to younger people on a voluntary basis. This advice was strengthened after the latest review last week.
However, despite the health ministry's new instructions, Italy’s regions are continuing to implement their own vaccine programmes with varying rules.

Italian news agency Ansa reported “chaos in the regions” on Tuesday, saying the latest rule-change on AstraZeneca had "caused confusion”.
Some Italian regional authorities said they will still continue to allow people to take AstraZeneca if they want to amid concerns about the supply of other vaccines, while Campania said it was banning the use of the Astrazeneca and Johnson and Johnson vaccines altogether.
EXPLAINED: Why has Italy recommended the AstraZeneca vaccine for over-60s only?
Vincenzo De Luca, regional president of Campania, announced that he had written to the health minister saying his region was against giving mixed vaccinations and claiming that the practice "has not been trialled broadly on an international level".
NO AL MIX DI VACCINI
— Vincenzo De Luca (@VincenzoDeLuca) June 13, 2021
NO ALLA PRIMA DOSE DI VACCINI A VETTORE VIRALE
Ecco la mia dichiarazione 👇https://t.co/2yih6a3A1g
In an interview with La Stampa on Monday, Health Minister Roberto Speranza stressed that mixing vaccinations was known to be safe and effective, saying: "Our indications are peremptory and must be followed. It is not a political debate. It’s not the prime minister, ministers or a regional president who decides:”
"So-called 'vaccination crossing' is something Germany has been doing for two months, which France and Spain have also been doing for some time,” he said. “It's not made up, these are scientific studies. This is a procedure that has given good results.”
Several other EU countries including Sweden, Norway and Austria are also allowing people to have an mRNA shot for their second dose if they were first vaccinated with AstraZeneca, after those countries also changed their advice on the jab.
READ ALSO: Is Italy really going to offer vaccines to tourists this summer?
The European Medicines Agency (EMA) has stressed that the AstraZeneca jab remains approved for all adults, saying the benefits continue to outweigh the risks.
Misinformation is making the rounds today. This is the situation: Benefit/risk balance of AstraZeneca #COVID19 vaccine is positive and it remains authorised for all populations.
— EU Medicines Agency (@EMA_News) June 13, 2021
Meanwhile the Italian region of Puglia said it would allow people to take AstraZeneca if approved by their doctor, and Lombardy’s regional government expressed concerns about the supplies of Pfizer and Moderna vaccines.
Several regions have found themselves with unused doses of the Oxford University vaccine, as people opt to get vaccinated with alternatives that they perceive as safer or that have a shorter interval between doses (or in the case of Johnson & Johnson, require just one shot).
As a result, some regions had been offering the Covid-19 vaccine to younger adults as part of vaccination “open days”: special vaccination drives, often at evenings or weekends, that allow the youngest age groups to get a shot earlier than they otherwise might.
The latest review of the use of AstraZeneca in Italy came following the news that an 18-year-old in north-west Italy who was vaccinated with AstraZeneca two weeks previously had died after developing a blood clot.
Most of the AstraZeneca side effects observed so far occurred within two weeks of the first dose. The second dose is thought to carry an even lower risk, though more data is needed to know for sure.
Data from millions of people vaccinated with AstraZeneca across Europe and the UK indicates that serious side effects after either dose remain extremely rare.
In a monitoring report released this week, AIFA said that in Italy the incidence of blood clots after vaccination was around one per 100,000 injections of AstraZeneca, mainly in people under 60.
It has not received reports of any clots developing after the second dose, the agency said.
What are the different types of Covid-19 vaccine available?
AstraZeneca Johnson & Johnson, both so-called 'viral vector' vaccines, are authorised for everyone over 18 in Italy and by Europe’s medicines agency, but both have been dogged by reports of rare blood clots.
The EU and Italy have also authorised two mRNA vaccines: Pfizer/BioNTech and Moderna.
Viral vector vaccines like AstraZeneca and Johnson & Johnson use genetically-engineered version of a common-cold causing adenovirus as a "vector" to shuttle genetic instructions into human cells.
The mRNA or Messenger RNA genetic technology instead trains the body to reproduce spike proteins, similar to that found on the coronavirus.
When exposed to the real virus later, the body recognises the spike proteins and is able to fight them off.
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.